When Resistant Dextrin Beats MCC and When It Definitely Does Not
Resistant dextrin has evolved from being just an “invisible fiber” in beverages into a practical soluble corn fiber excipient for modern nutraceutical solids. The key question we frequently hear from formulators is straightforward: can resistant dextrin replace microcrystalline cellulose (MCC)—or at least share the workload as a filler, disintegration aid, or coating solid? Our view is balanced: resistant dextrin is best positioned as a clean-label, low-calorie, highly soluble functional ingredient that can support disintegration and film-coating systems. While it offers unique benefits in solubility and nutritional profile, it is not a one-for-one substitute for MCC regarding compaction reliability or for classic super-disintegrants in terms of speed.
What Resistant Dextrin Looks Like in Our Portfolio
At Shandong Shine Health, our resistant dextrin is produced from non-GMO corn starch using a specialized enzymatic process, focusing on consistent, food-grade quality. For buyers evaluating resistant dextrin as a tablet ingredient, the specifications that matter most during early screening are clear and simple:
Fiber content: ≥82%
Protein: ≤6.0%
Appearance: White to light yellow powder
Storage: Store in a cool, dry place
Functional profile: Fast dissolving, low viscosity, thermally stable, clean taste
This combination makes resistant dextrin attractive when the tablet itself is also expected to deliver nutritional value (fiber) and a clean-label story, rather than just serving as an inert carrier.
Resistant Dextrin vs. MCC for Tablet Performance
In tablet manufacturing, MCC remains the industry benchmark because it compacts well, flows reliably, and supports disintegration through wicking. Resistant dextrin behaves differently primarily because it is highly soluble. Understanding this distinction is crucial for formulation success.
Quick Comparison for Formulation Decisions
What you need | Resistant Dextrin | MCC (Microcrystalline Cellulose) |
Primary role | Soluble corn fiber excipient; nutritive filler | Insoluble filler-binder; compaction backbone |
Typical “breakup” behavior | Dissolution + pore formation (limited swelling) | Wicking + moderate swelling |
Best-fit products | Nutraceutical tablets, chewables, lozenges | Broad use in direct compression tablets |
Main risk to manage | Moisture pickup and softness if poorly packaged | Slower breakup if overdosed or over-lubricated |
Practical takeaway: Resistant dextrin allows you to reduce reliance on purely inert fillers and improve label appeal, while retaining MCC to keep mechanical properties predictable.
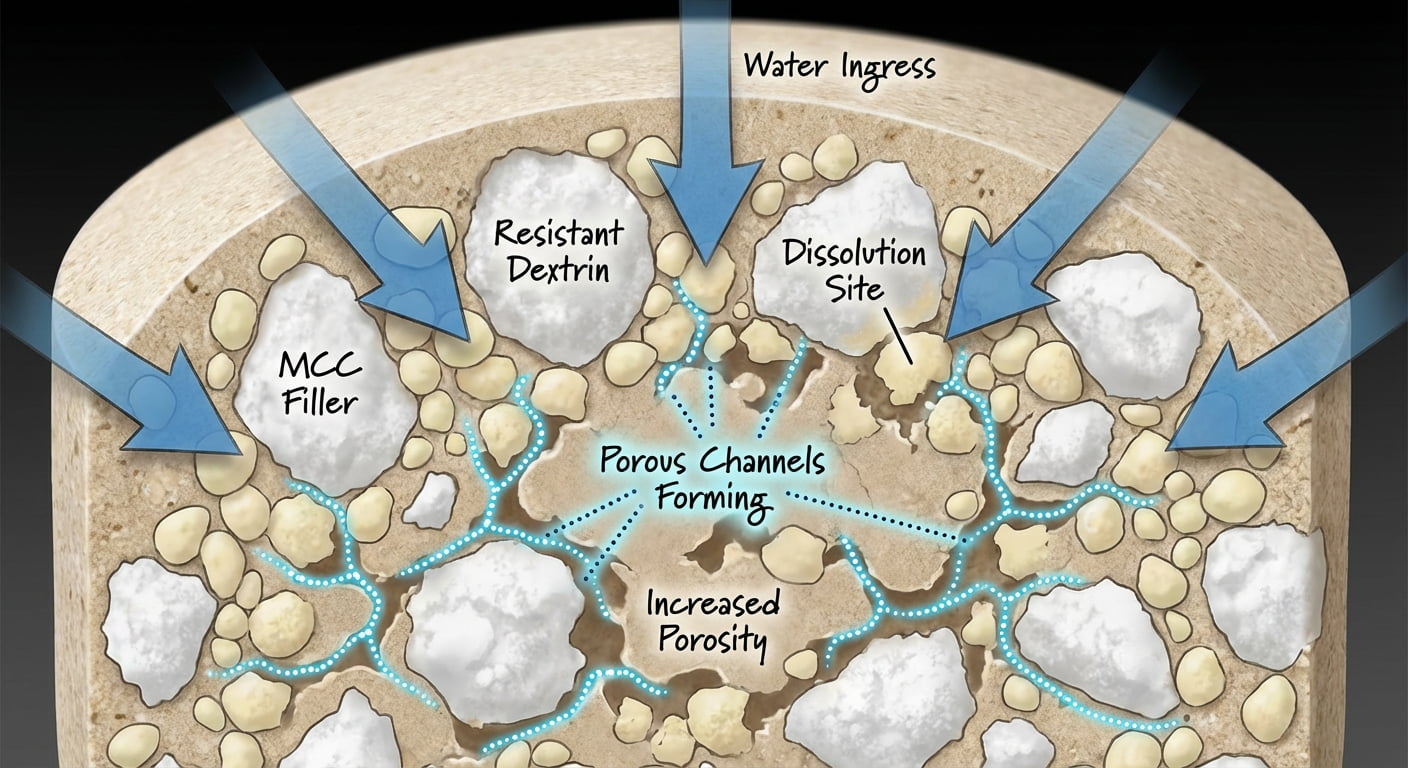
Can Resistant Dextrin Act as a Disintegrant?
A true disintegrant drives rapid water ingress and tablet breakup via capillary action, swelling, and internal stress. Because resistant dextrin is soluble, its contribution to disintegration is mainly through dissolving to create channels rather than aggressive swelling. This mechanism creates pathways for water to penetrate the tablet core.
Where resistant dextrin tends to work well:
Chewable tablets and pressed lozenges where fast mouth-dissolve affects consumer experience.
High-fiber nutraceutical tablets where resistant dextrin is part of the active nutrition story.
Lower-risk formulations that can tolerate longer disintegration times than pharmaceutical IR standards.
Where resistant dextrin is usually not enough by itself:
Fast-disintegrating swallow tablets that must hit very short disintegration times.
Systems that depend heavily on swelling-based matrix disruption.
If you require rapid breakup, resistant dextrin can still be valuable—but typically works best alongside MCC and/or a super-disintegrant.
How Resistant Dextrin Fits in Tablet Coatings
Formulators exploring fiber-forward coatings are often surprised by how useful resistant dextrin can be in aqueous systems. Because it is water-soluble and low viscosity, it can function effectively as:
A co-film former (supporting solids in the coat).
A nutritive coating solid for “fiber-enriched” positioning.
A mouthfeel and appearance contributor for chewables and nutrition tablets.
However, it is not designed to be a high-performance moisture barrier; its solubility makes coats more “breathable,” which is advantageous for dissolution but requires careful consideration for moisture-sensitive actives.
A Simple R&D Screen to Compare Resistant Dextrin and MCC
For an efficient resistant maltodextrin tablet formulation trial, we recommend a quick set of head-to-head screens (keeping all other variables constant):
Water uptake / dissolution behavior (Check at 15/30/60 minutes).
Wicking test (Using powder bed or strip method).
Compression screen with 0%, 5%, 10%, and 20% resistant dextrin replacing your filler fraction.
Humidity challenge (Watch for softening and sticking; this helps define your packaging and desiccant strategy).
Coating sprayability with small additions of resistant dextrin as a solids partner.
Supplier Checklist for Sourcing
If you are searching for a reliable manufacturer, use a checklist that goes beyond price to ensure formulation success:
Full COA: Check fiber %, protein, moisture, heavy metals, and microbiology.
Traceability: Verify non-GMO corn starch origin and batch records.
Production controls: Look for automated processing, in-house QC testing, and GMP workshop standards.
Certifications: ISO9001, BRC, HALAL, HACCP, KOSHER (as applicable).
Application support: Ask for blend guidance, stability considerations, and sample availability.
We manufacture resistant dextrin on a controlled process designed for consistent performance as a soluble corn fiber excipient, and we support customers who benchmark resistant dextrin directly against MCC in their own tablet lines.
Contact Us
Ready to test resistant dextrin in your next formulation? Reach out to us for samples or a full COA.
Email: info@sdshinehealth.com
WhatsApp: +86 13405443339
Data Sources
Lopes, A. P., et al. (2017). Production of gastro-resistant coated tablets. African Journal of Pharmacy and Pharmacology.
United States Pharmacopeia. (2024). General Chapters on tablet disintegration and dissolution.
ICH. (2003). Q1A(R2) Stability Testing of New Drug Substances and Products.
FDA. (2023). Dietary supplements—manufacturing and quality considerations.
Aulton, M. E., & Taylor, K. (2018). Aulton’s Pharmaceutics: The Design and Manufacture of Medicines.