Stop Tablet Variability With Microcrystalline Cellulose Quality Control
See how Shandong Shine Health runs microcrystalline cellulose quality control end to end, linking GMP in-process controls to MCC CoA specs and tablet performance.
Microcrystalline cellulose (MCC) often looks like a simple white powder, but anyone running direct compression knows the truth: small shifts in moisture, particle size distribution, or density can quickly turn stable production into sticking, capping, or weight variation. This is why microcrystalline cellulose quality control must be built into manufacturing—not left to a final lab report.
Where Microcrystalline Cellulose Quality Control Is Won or Lost
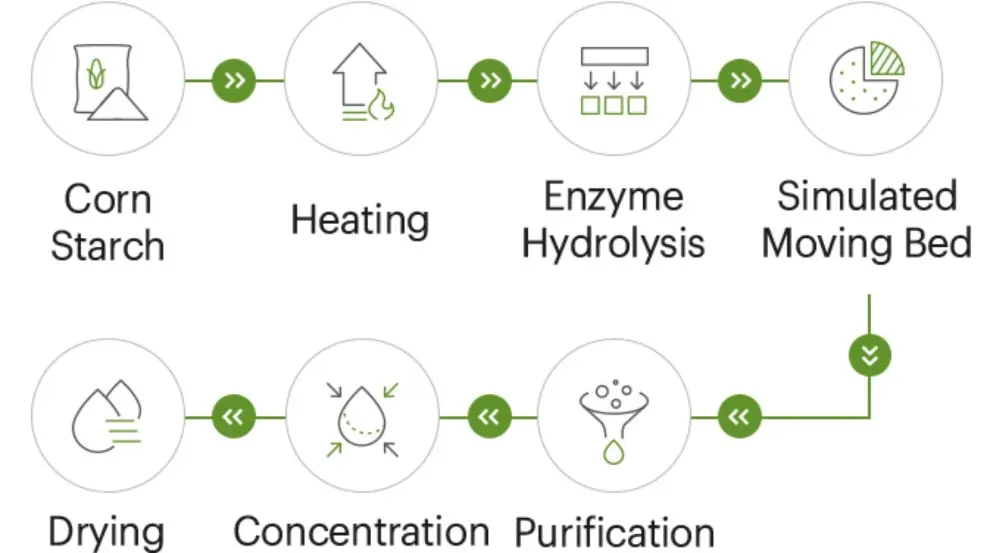
At Shandong Shine Health, microcrystalline cellulose quality control is managed as an end-to-end system in a GMP workshop—from purified cellulose acceptance to the final bag release. Our automated production lines (German origin) and standardized procedures help keep variability low, but the real value comes from consistent controls at every stage of the MCC manufacturing process.
Step 1: Raw Material Acceptance and Traceability
We start from purified cellulose with full supplier documentation and lot traceability. Before any material enters production, microcrystalline cellulose quality control checks typically focus on identity, appearance, and potential contaminants (including ash and visible impurities where applicable). This step prevents avoidable downstream issues that can later show up as poor flow or unexpected compaction behavior.
Step 2: Hydrolysis Washing and Neutralization Controls
Controlled acid hydrolysis builds the crystalline structure that makes MCC a reliable pharmaceutical excipient. In this stage, microcrystalline cellulose quality control is driven by rigorous in-process monitoring:
- Time and temperature profiles to maintain consistent depolymerization.
- pH or conductivity checks during washing to confirm residual acid removal.
- Validated rinse cycles for repeatable cleanliness.
These controls support microcrystalline cellulose quality control by reducing the risk of residual acidity, which can affect compatibility with sensitive actives or lead to stability concerns.
Step 3: Drying Control—Keeping Moisture in the Right Window
Drying is one of the most sensitive points for microcrystalline cellulose quality control. Over-drying can increase dusting and brittleness; under-drying can reduce flow and worsen storage stability.
We routinely manage:
- Loss on drying (LOD) targets commonly aligned with compendial practice (many MCC suppliers target ≤7%).
- Moisture trending, using SPC-style monitoring to catch drift early.
- Water activity checks when required by customer or application needs.
Maintaining a stable moisture window helps keep microcrystalline cellulose quality control tied to what matters on your press: consistent hardness, fewer defects, and predictable disintegration.
Step 4: Milling, Sieving, Blending, and Grade Consistency
For direct compression, particle engineering is performance engineering. Here, microcrystalline cellulose quality control focuses on:
- Particle size distribution (PSD), commonly tracked via D10/D50/D90.
- Sieve integrity and metal detection.
- Bulk and tapped density, which are critical for die fill and tablet weight.
Blending is also part of microcrystalline cellulose quality control—it helps ensure that performance is consistent from bag to bag, especially for customers running multiple compression lines.
What to Verify on an MCC CoA
If you are evaluating manufacturers for your supply chain, the Certificate of Analysis (CoA) is where the supplier’s microcrystalline cellulose quality control becomes visible.
A practical CoA review should include:
- Identity confirmation (commonly IR).
- LOD and residue on ignition/ash.
- PSD and density for the exact MCC grade used.
- Microbial limits and other safety indicators as required.
- Clear references to USP/EP or other compendial methods.
Below is a simplified, indicative view of common grades used in tableting; always confirm your batch values on the CoA:
| MCC grade | Typical use | Key CoA focus for microcrystalline cellulose quality control |
|---|---|---|
| PH-101 | Strong binding, fine particle | PSD window, LOD, density consistency |
| PH-102 | Improved flow vs PH-101 | Flow/packing (PSD), bulk/tapped density |
| PH-200 | Coarser grade for higher flow | Coarse PSD control, die fill consistency |
Lab Sampling, Retention, and Trend Monitoring
A robust lab and sampling system turns microcrystalline cellulose quality control into repeatable outcomes:
- Lot-based sampling plans for release decisions.
- Retention samples kept for each batch (traceability and complaint investigation).
- Routine tests for MCC: identity, LOD, PSD, bulk/tapped density, and microbiology.
By trending key parameters (such as LOD and D50), microcrystalline cellulose quality control can identify drift before it becomes an out-of-spec event.
Why MCC Specs Directly Affect Tablet Performance
From a formulation standpoint, microcrystalline cellulose quality control protects three outcomes: flow, compaction, and disintegration.
- Moisture (LOD): Levels that are too high can worsen sticking and capping, while levels that are too low can drive friability.
- PSD (D10/D50/D90): Influences blend uniformity, packing, and weight control.
- Bulk density: Impacts tablet weight and thickness at a fixed die volume.
We recommend confirming microcrystalline cellulose quality control with quick internal checks during qualification (small-scale compression, hardness/friability, and disintegration).
One Supplier, Two QC Workflows: MCC and Resistant Dextrin
Many customers source both tablet excipients and functional fibers from one partner. We manufacture MCC and resistant dextrin in the same GMP environment, but microcrystalline cellulose quality control is distinct from resistant dextrin quality control.
- Microcrystalline cellulose quality control: Focuses on identity, LOD, PSD, density, microbial safety, and performance-related consistency.
- Resistant dextrin quality control: Focuses on fiber content (commonly ≥82% for our resistant dextrin grades), moisture, ash, protein, water activity, and microbiology.
If you are evaluating a Recommended Chinese Resistant Dextrin Manufacturer, ask to see how the dextrin CoA aligns with your food or supplement application requirements.
Next Steps for Buyers
If your priority is stable direct compression and fewer batch-to-batch surprises, start by benchmarking your supplier’s microcrystalline cellulose quality control against your tablet performance needs.
To request recent CoAs or qualification samples, contact Henry Liu at info@sdshinehealth.com or message us on WhatsApp.
References
- United States Pharmacopeia. (n.d.). Microcrystalline Cellulose (excipient resource). https://www.usp.org/harmonization-standards/pdg/excipients/microcrystalline-cellulose
- Thoreens, G., Krier, F., Leclercq, B., Carlin, B., & Evrard, B. (n.d.). Microcrystalline cellulose, a direct compression binder in a quality by design environment—A review. http://www.phexcom.com/UploadFiles/…
- Miljković, V., Nikolić, L., & Miljković, M. (2024). Microcrystalline cellulose: A biopolymer with diversiform applications. Cellulose Chemistry and Technology. https://doi.org/10.35812/cellulosechemtechnol.2024.58.62
- Hindi, S. S. Z. (2017). Microcrystalline cellulose: The inexhaustible treasure for pharmaceutical industry. Nutrition and Research. https://doi.org/10.12691/NNR-4-1-3
- Heng, P. W. S., Liew, C. V., & Soh, J. L. P. (2004). Pre-formulation studies on moisture absorption in microcrystalline cellulose. Chemical & Pharmaceutical Bulletin, 52(4), 384–388. https://doi.org/10.1248/CPB.52.384
- Chaerunisaa, A. Y., Sriwidodo, & Abdassah, M. (2019). Microcrystalline cellulose as pharmaceutical excipient. In Pharmaceutical Formulation Design—Recent Practices. https://doi.org/10.5772/INTECHOPEN.88092